
A better way to secure data: Introducing Federated SSO to benefit Hospital IT
Implementing Federated Single Sign-On (SSO) simplifies authentication for MDIP users. This streamlined experience helps reduce login fatigue.

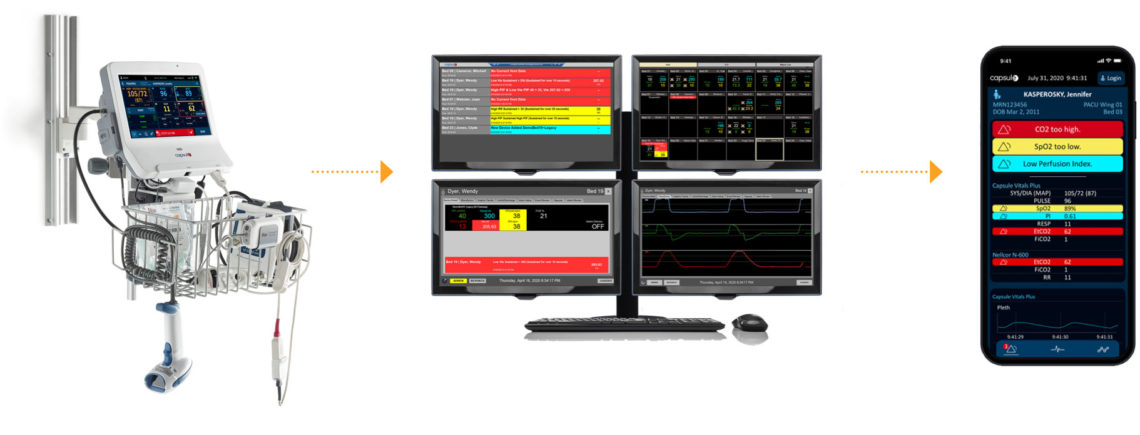
Vitals Plus patient monitoring, now with capnography, is part of the Capsule Medical Device Information Platform (MDIP), a solution that enables hospitals to simplify workflow and enhance caregivers’ ability to deliver quality, safe care. A well-organized, vivid display of patient vitals and an integrated Early Warning Scoring System (EWSS) that calculates a score quickly at the bedside (combining certain vitals and clinician observations), help to identify patients at risk of deterioration. More efficient workflows and easy acquisition of vital signs, and the ability to continuously monitor critical parameters, translates to more time with the patient and helps ensure that the correct patient vital signs information is incorporated into the patient record.
Added measurements expand vitals acquisition and utilization:

The Capsule Early Warning Scoring System* is an optional patient monitoring application that uses vital signs data acquired through Capsule Vitals Plus. Simple to use, automated and reliable, the Early Warning Scoring System** registers subtle changes in the patient’s state while the clinician is at the bedside and able to intervene. EWSS is configured by each hospital to provide an immediate aggregate score, and this score can guide clinicians through appropriate actions as defined by hospital protocol. This includes sending the score to alarm and alert systems that can activate a rapid response or medical emergency team. The Capsule Early Warning Scoring System also provides detailed trending information to place patients’ scores in clinical context.
*Early Warning Scoring System (EWSS) is a hospital-configured medical calculator.
**Not available on Chart Xpress outside the U.S. and Singapore.
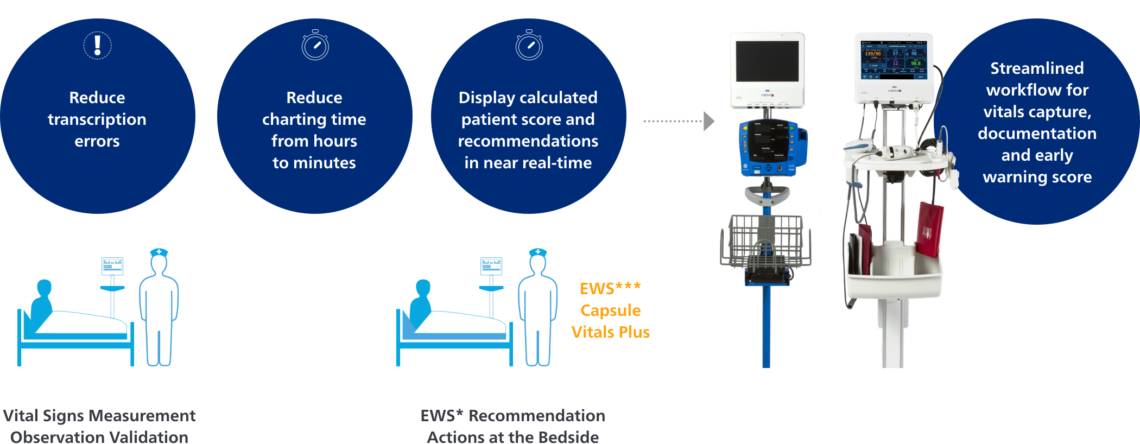

An all-in-one solution for patient monitoring and clinical documentation that combines ease-of-use and efficiency.
Download
Implementing Federated Single Sign-On (SSO) simplifies authentication for MDIP users. This streamlined experience helps reduce login fatigue.

EDM-enabled data is easier to use across a health system’s ecosystem, whether data is feeding into an EMR, central station, clinical decision support system, or an analytics platform.