Clinical Insights Manager
Together, we can unlock your data and put it to work
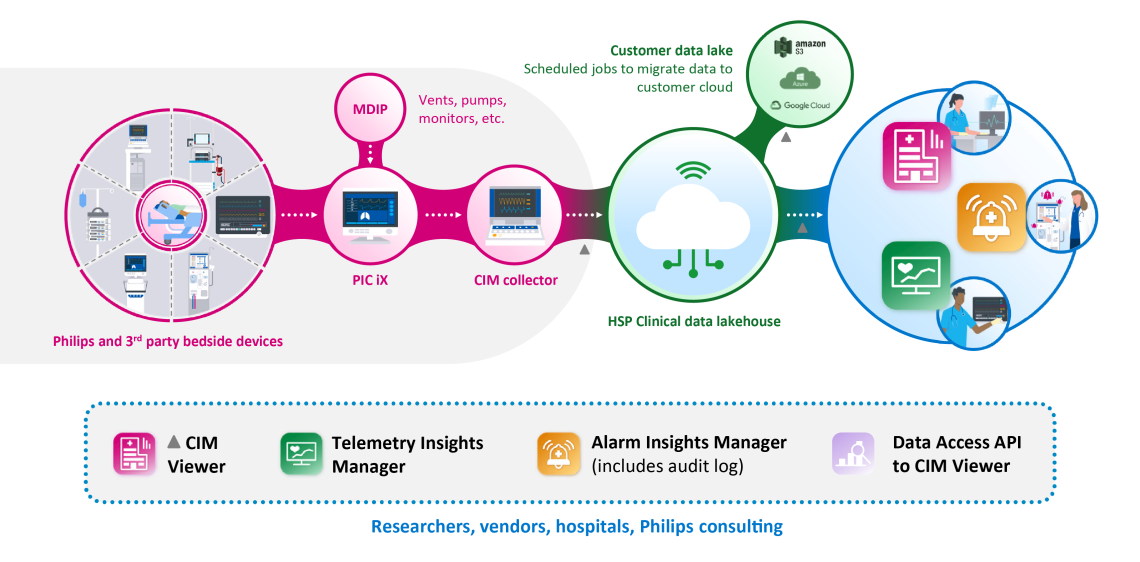
Clinical Insights Manager (CIM) is Philips’ new cloud-based (Health Suite Digital Platform), service-enabled, enterprise-wide platform that allows hospitals to capture, store, and analyze high-fidelity patient data utilizing tools and steps towards quality improvement, alarm management, research initiatives and operations management.
Philips Clinical Insights Manager Solutions is the only service-oriented, end-to-end solution enabling high-resolution physiologic data acquisition, archiving, and access. Designed to protect your data and security, Clinical Insights Manager enables a secure offering that further fortifies controls to better withstand cyberattacks and complies to global privacy regulations.
How the Clinical Insights Manager solution stands out:
- An open and scalable cloud platform that aggregates, stores, and filters high-resolution retrospective data (including diagnostic-quality ECG) through tools and intuitive dashboards to easily navigate and query data sets.
- Ensure data quality and integrity to confidently leverage insights, from both Philips and 3rd party devices, to improve alarm fatigue, achieve KPI’s, enhance caregiver workflow and reduce noise in critical care units to improve patient care & outcomes.
- Philips Clinical Professional Services helps execute quality and research initiatives, creates sophisticated algorithm development and clinical process optimization, along with monitoring customers’ progress against their established goals.
Alarm Insights Manager
- Provides visualization of an intuitive set of alarm key performance indicators (KPIs) to assess alarm burden over hours, days, weeks or months, and by clinic or unit, or by patient or alarm type.
- Identifies actionable insights into hospital alarm system quality through queries, aggregation and filters, to provide insights into the root cause of alarm fatigue, improve alarm accuracy and severity.
- Allows for historical benchmark comparisons across units, months, weeks and by specific KPls for continuous improvement.
- Provides data surrounding alarm, alert and event information, including technical alarms, event onset time, alarm announcement time and text and alarm silence times.
- Provides a one-time migration of the Philips PIC iX Alarm Audit Log (maximum 90 days) to Alarm Insights Manager’s Alarm Audit Log Migration (AALM) tool.
- Ability to create and save custom reports as well as automate and share reports with stakeholders.
Telemetry Insights Manager
- Visibility of Telemetry Equipment Inventory: Track the status of telemetry devices, whether assigned, available, or dormant, to ensure optimal utilization and readiness.
- Arrhythmia Severity Alarm Data: Access detailed information on arrhythmia severity alarms to evaluate telemetry usage and enhance patient monitoring.
- Patient Monitoring Status: Identify patients who have active telemetry orders in the Hospital Information System (HIS) but are not currently monitored, and vice versa.
- Time on Telemetry Evaluation: Analyze the duration of patients’ time on telemetry to support healthcare management, quality control, and resource utilization.
- Daily Management and Reporting: Gain timely access to data for daily management tasks and shift change reporting activities.
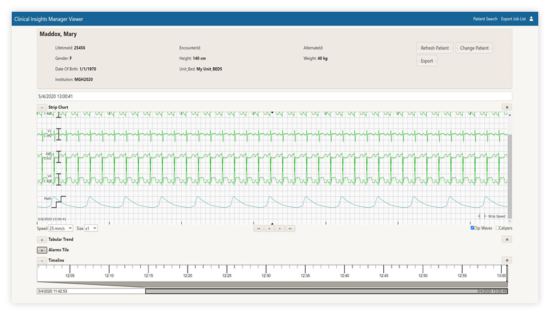
Clinical Insights Manager Viewer
- Access retrospective patient-specific data over one year of prior history.
- Comprise complete patient information such as demographics, waveforms, numeric and alarms – in one intuitive view.
- Ability to search, review, and extract patient data (including the ability to de-identify data).
- Patient Data Auto Export – Multi Cloud Integration allows scheduling of export jobs as a predefined frequency and can integrate with a facility owned and managed cloud storage account such as Google Cloud Storage, AWS S3 or Azure Blob storage.
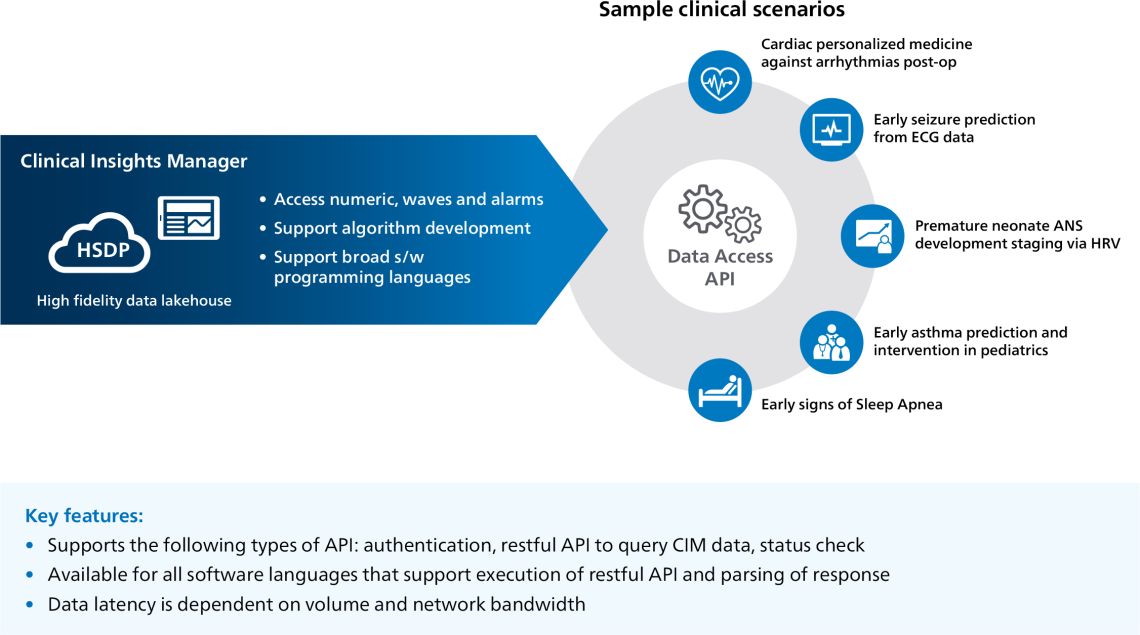
- Data Access API provides Enhanced Ql/research support through fast and easy data migration including improved data quality.
- Aligns all data (alarms, numeric, and waveform) based on an event.
- Allows the user to export a single patient or multiple patient data to the user’s local repository.
Data Access API supports algorithm development for a broad range of clinical use cases

Privacy/security
- Option to de-identify on-premises or upon cloud export
- Security assessment according to NIST-800-53 controls
- Security and vulnerability scans
- Penetration and exploitability testing to find security threats that may come from malicious outsider or insider user of CIM

Data communication
- Data at rest in cloud is encrypted using AES 256 RSA
- Data at rest on premise is encrypted using AES 256 RSA
- All communication uses TLS V1.3
- All communication to the applications are via HTTPS only

Network and cloud
- Microservices are deployed in a Virtual Private Cloud within HealthSuite
- API Gateway Architecture
- All communication to and within Cloud Microservices is encrypted with HTTPS

Application
- HSDP OAuth authenticates each individual uniquely for access to Web Applications
- Multi-factor authentication enabled
- HTTPS request requires web token issued against an identity management service with short lifetime validity
Are you ready to advance
your care delivery?